|
Interaction of X-rays with MatterĪlthough a large number of possible interactions are known, there are three key interaction mechanisms with matter. The quantity h/m ec is known as the Compton wavelength of the electron and is equal to 2.43×10 −12 m. The maximum change in wavelength can be derived from Compton formula: In this case the photon transfers to the electron as much momentum as possible. The maximum change in wavelength ( λ′ − λ) for the photon occurs when Θ = 180° (cos(Θ)=-1). The minimum change in wavelength ( λ′ − λ) for the photon occurs when Θ = 0° (cos(Θ)=1) and is at least zero. Where λ is the initial wavelength of photon λ’ is the wavelength after scattering, h is the Planck constant = 6.626 x 10 -34 J.s, m e is the electron rest mass (0.511 MeV) c is the speed of light Θ is the scattering angle. The shift of the wavelength increased with scattering angle according to the Compton formula: So the decrease in photon’s momentum must be translated into decrease in frequency (increase in wavelength Δ λ = λ’ – λ). This deflection results in a decrease in energy (decrease in photon’s frequency) of the photon and is called the Compton effect. In Compton scattering, the incident gamma-ray photon is deflected through an angle Θ with respect to its original direction. As a result of momentum conservation law, the photon must lower its momentum given by: While the electron gains momentum (mass x velocity), the photon cannot lower its velocity. Upon collision, the photon bounces off electron, giving up some of its initial energy (given by Planck’s formula E=hf). In the case of Compton scattering the photon of frequency f collides with an electron at rest. Compton scattering formula is the mathematical relationship between the shift in wavelength and the scattering angle of the X-rays. Compton explained that the X-ray shift is caused by particle-like momentum of photons. Possible implications of the production process of the hydride are briefly discussed.The Compton formula was published in 1923 in the Physical Review. The results indicate that the ionic picture of MgH2 is not simple since its outer electrons are extended. Both calculations yielded profiles which are in fairly good agreement with the experimental isotropic profile. In the second approach, the Compton profile of MgH2 was calculated using the tight-binding linear combination of atomic orbitals (LCAO) method, including large overlaps. In the first approach, the Compton profile of the valence electrons in MgH2 was calculated by the pseudopotential orthogonalized-plane-wave (OPW) method, including core orthogonalization. Two different theoretical approaches have been used for comparison with the experimental data. 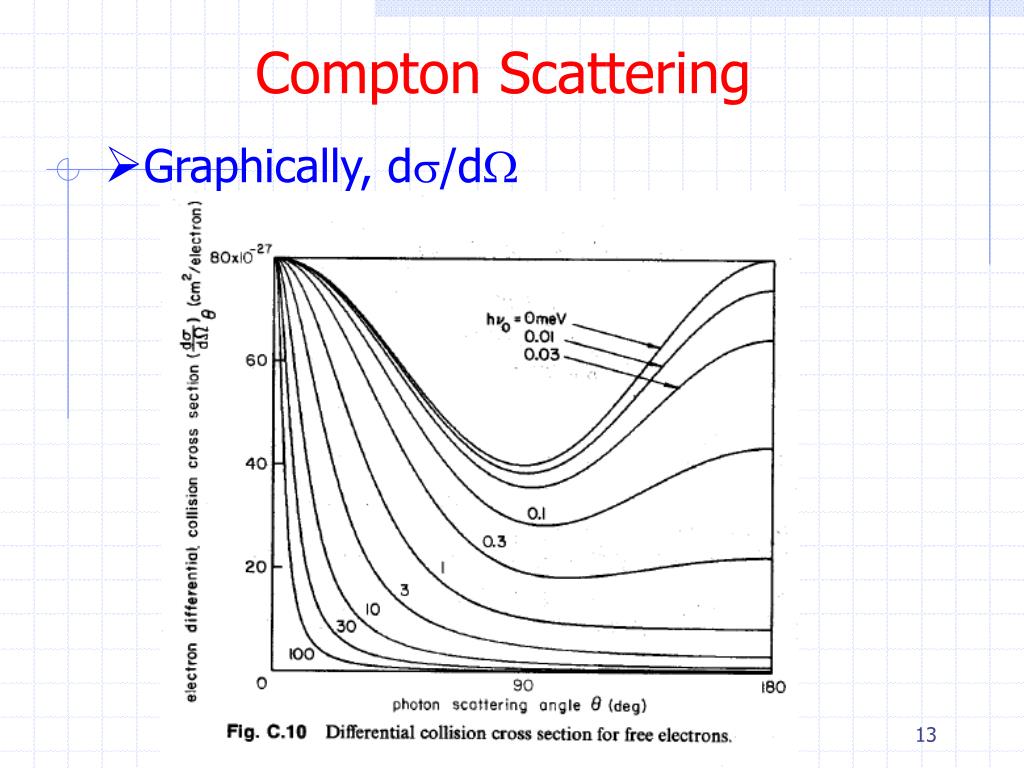
Possible implications of the production process of the hydride are briefly discussed.ĪB - Compton scattering measurements have been made on magnesium hydride (MgH2) using the 59.54-keV gamma rays from a 300-mCi Am241 source at a scattering angle of 150°. N2 - Compton scattering measurements have been made on magnesium hydride (MgH2) using the 59.54-keV gamma rays from a 300-mCi Am241 source at a scattering angle of 150°. T1 - Compton scattering study of the electronic structure of magnesium hydride
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
